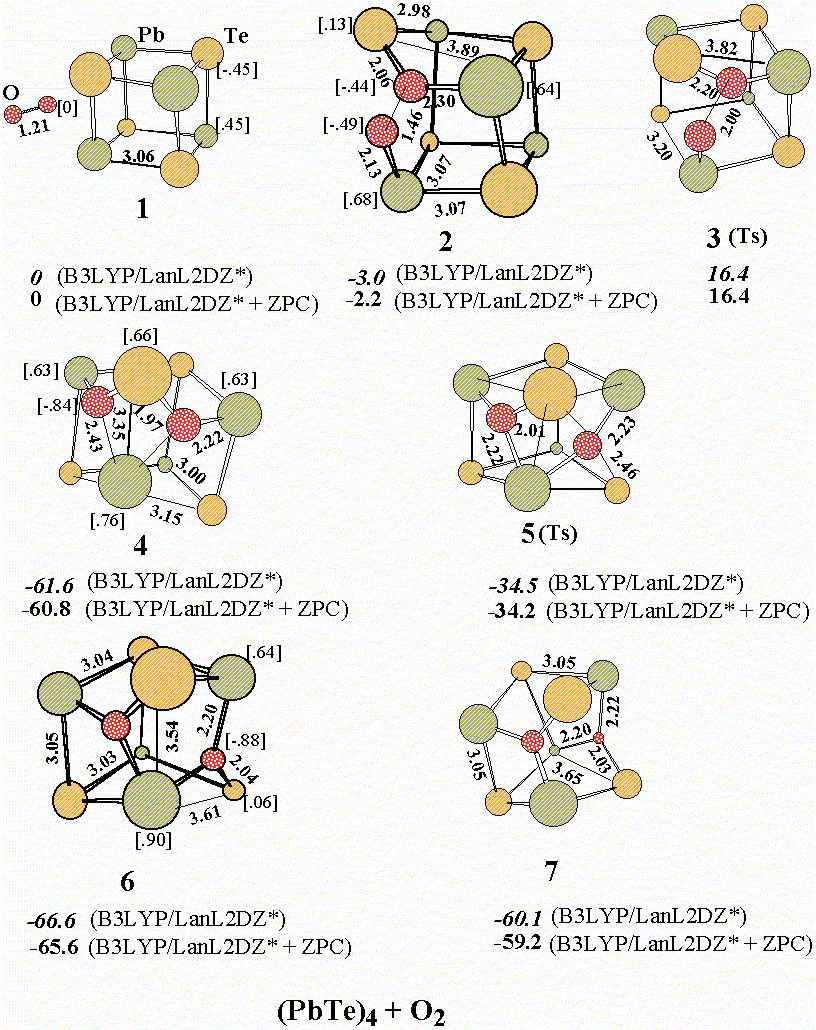Joint Semiconductors Surface Study group: research sketches
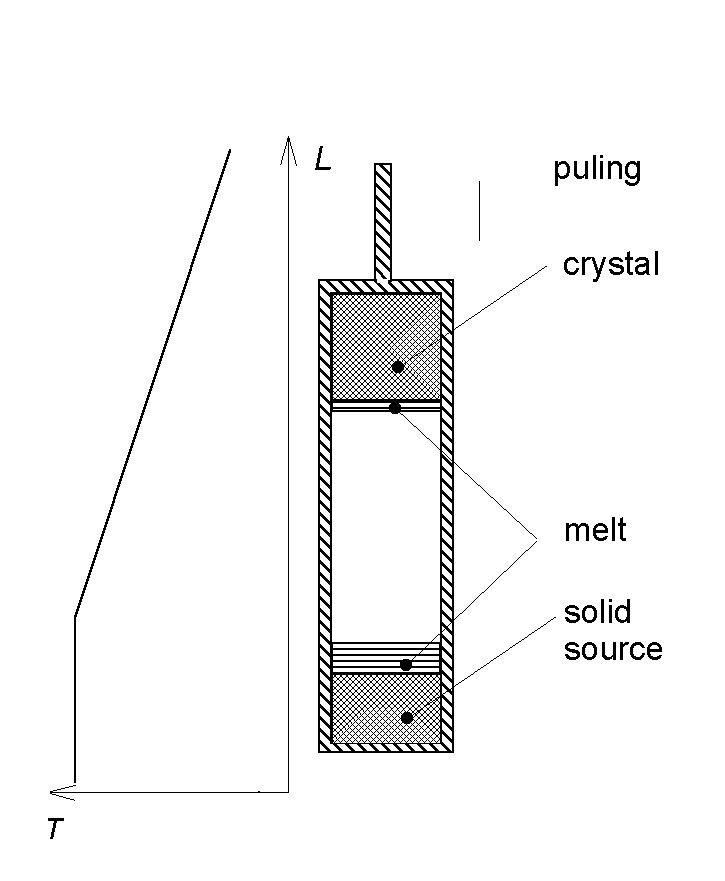
The Joint Semiconductors Surface Study (JSSS) group was developed on the base of Inorganic Chemistry division, Chemistry dept. of M.V. Lomonosov Moscow State University. It includes also specialists from three different academician institutes RAS. Our group is aimed at studying the surface properties and reactivity of A4B6 semiconducting materials and their solid solutions. They are widely used in IR optoelectronics and for thermoelectrics development. The experiments are carried out using single crystals, which are obtained at MSU by different growth techniques, namely, the Bridgman and the VLS methods. In order to obtain single crystals being both structurally and compositionally perfect, one has to know precisely the phase diagrams of the systems under study. At MSU the binary, ternary and quasiternary phase diagrams of A4B6-based systems are also studied using thermal analysis techniques.
The schematic sketch of VLS growth using 3-phase source.
The surface analysis techniques used for the investigations are various spectroscopic (XPS, AES, AR XPS, UPS, EELS), microscopic (STM) and diffraction (LEED) methods. Most of the experiments are performed using ESCALAB MKII and ESCALAB 5 spectrometers.
The main research topics of surface studies are: